UV-Visible Spectroscopy
Introduction
UV-Visible spectroscopy is a fundamental analytical technique used to measure the absorption of ultraviolet and visible light by chemical substances. By analyzing how molecules interact with electromagnetic radiation in the 200–800 nm range, researchers obtain critical information about molecular structure, concentration, and electronic transitions. This technique is widely adopted in chemistry, biotechnology, pharmaceutical sciences, environmental analysis, and materials research due to its reliability, speed, and quantitative precision.
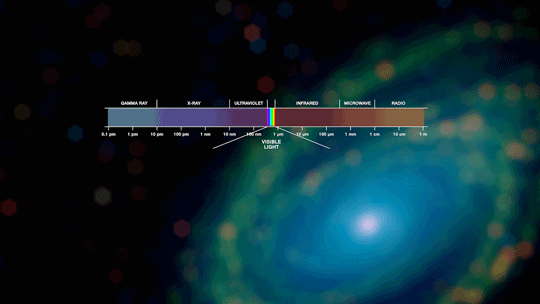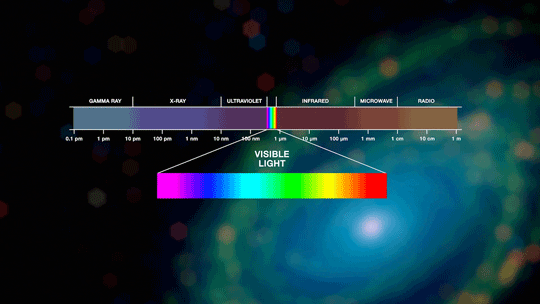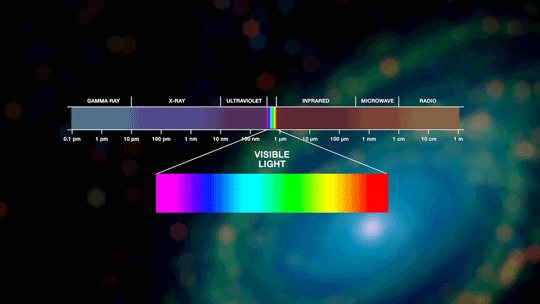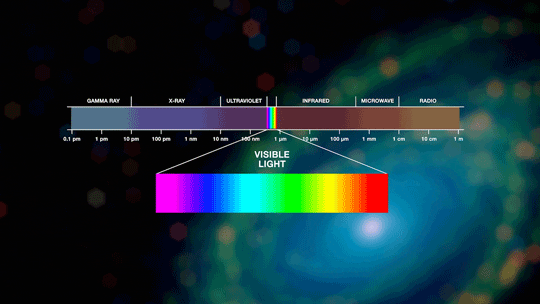
Principle of UV-Visible Spectroscopy
UV-Visible spectroscopy is based on the absorption of light energy that promotes electrons from lower to higher energy states. When a beam of light passes through a sample, specific wavelengths are absorbed depending on the molecular structure. The amount of absorbed light is mathematically described by the Beer–Lambert Law: A = εcl Where:
- A — Absorbance
- ε — Molar absorptivity coefficient
- c — Concentration of the analyte
- l — Optical path length
This relationship enables precise quantitative analysis of dissolved compounds.
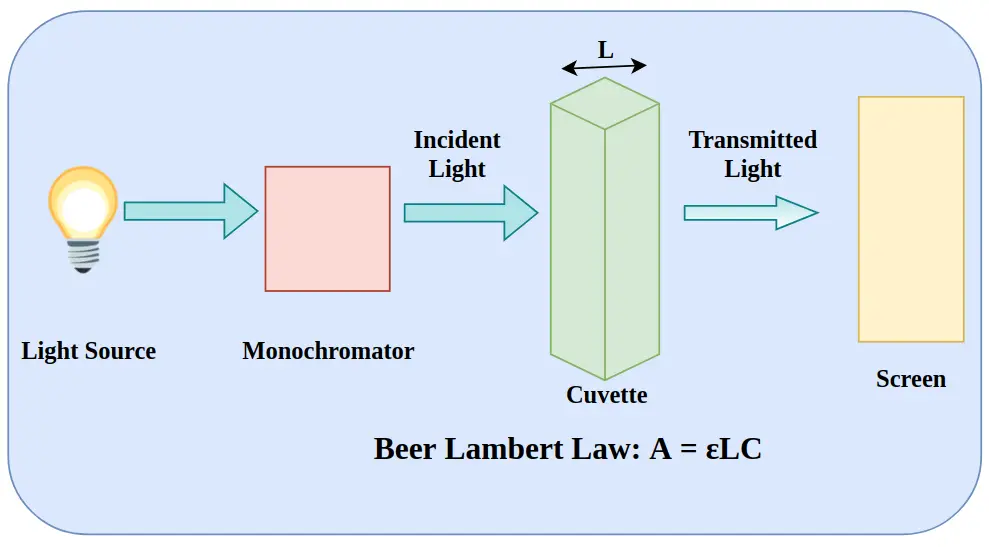
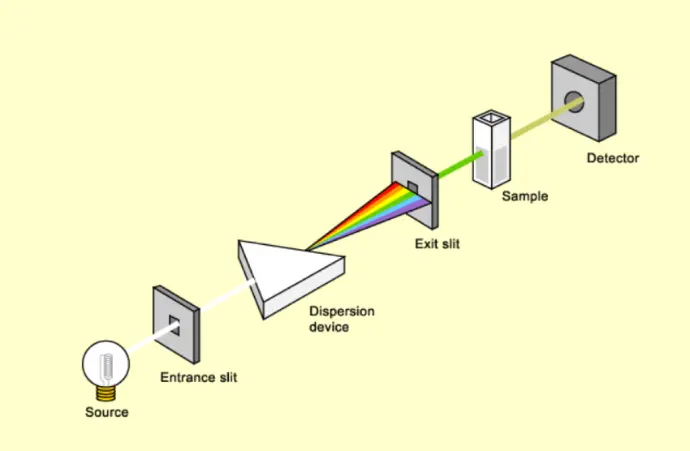
Instrumentation Components
Light Sources UV lamps (deuterium) and visible lamps (tungsten-halogen) provide stable radiation across the spectral range.
Monochromator Isolates specific wavelengths for accurate spectral measurement.
Sample Holder (Cuvette System) Transparent cuvettes maintain defined optical path length and chemical compatibility.
Detector Photodiodes or photomultiplier tubes convert transmitted light into measurable electrical signals.
Data Processing Unit Software systems convert signals into absorbance spectra and quantitative outputs.
Scientific Applications
UV-Visible spectroscopy supports research and industry in multiple domains:
Analytical Chemistry Quantitative determination of solute concentrations and reaction monitoring.
Pharmaceutical Research Drug quantification, purity testing, dissolution profiling, and formulation studies.
Biotechnology & Life Sciences DNA/RNA quantification, protein concentration measurement, and enzyme kinetics.
Environmental Analysis Water quality testing and detection of organic pollutants.
Materials Science Optical characterization of polymers, coatings, and nanomaterials
Advantages of UV-Visible Spectroscopy
- Rapid and non-destructive analysis
- High quantitative accuracy
- Simple sample preparation
- Cost-effective operation
- Suitable for routine and advanced research
Limitations and Considerations
- Requires optically transparent samples
- Spectral overlap may affect selectivity
- Lower structural detail compared to advanced spectroscopic methods
- Sensitive to stray light and instrument calibration

Data Interpretation and Quantification
UV-Vis spectra provide characteristic absorption peaks corresponding to molecular electronic transitions. Peak intensity correlates with concentration, while wavelength position indicates molecular structure. Quantitative workflows typically include:
- Calibration curve generation
- Blank correction
- Baseline normalization
- Replicate measurements for reproducibility
Conclusion
UV-Visible spectroscopy is a cornerstone analytical method for molecular detection and quantitative chemical analysis. Its balance of precision, simplicity, and speed makes it indispensable across scientific disciplines. LabSpectrix supports laboratories with precision instrumentation designed for reliable and reproducible spectroscopic analysis.